 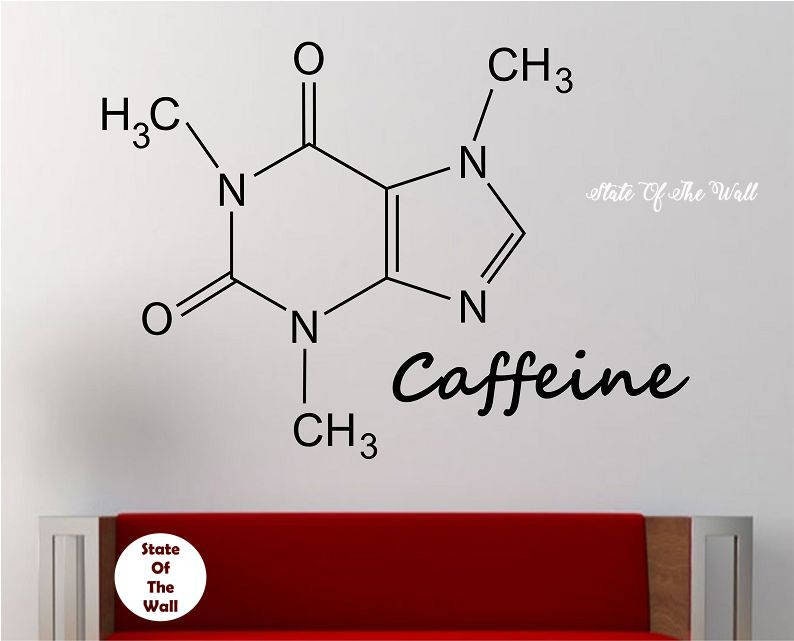
The food industry uses the caffeine as additive in many drinks like cola, tea or infusions. It is also used largely around the world as drug such as analgesics. Uses: Caffeine is highly consumed in the century XXI societies and most of the countries around the world. The caffeine molecule can act in this way because the molecule structure is highly similar to adenosine molecule, particularly on the part corresponding to the nitrogen base adenine. 
It is suspected the mechanism of action involved the reversibly blocks the action of adenosine in some receptors and thus, it is stimulated the nervous system. It is soluble in water.Ĭhemical properties: Caffeine is a central nervous system stimulant. Caffeine tastes bitter and the density is 1.23 g mL -1 and its melting point is 235 ✬ and at higher temperatures, it decomposes. Caffeine Formula: C8H10N4O Molecular weight: 194.1906 IUPAC Standard InChI: InChI1S/C8H10N4O2/c1-10-4-9-6-5(10)7(13)12(3)8(14)11(6)2/h4H,1-3H3 Copy IUPAC. Physical properties: Caffeine is a white, odorless and hygroscopic crystalline solid. High caffeine levels have also been found in the surrounding soil of coffee bean seedlings. It is a xanthine alkaloid compound that acts as a psychoactive stimulant. from a caffeine molecule for which its affinity is 10,000 times weaker. There are a few methods to prepare caffeine in chemical laboratories these methods include the reaction between dimethylurea and malonic acid. To View the Caffeine Molecule ->in 3D with Jmol About Caffeine. inhibitor effect Antisense antidote No them rather special molecules. 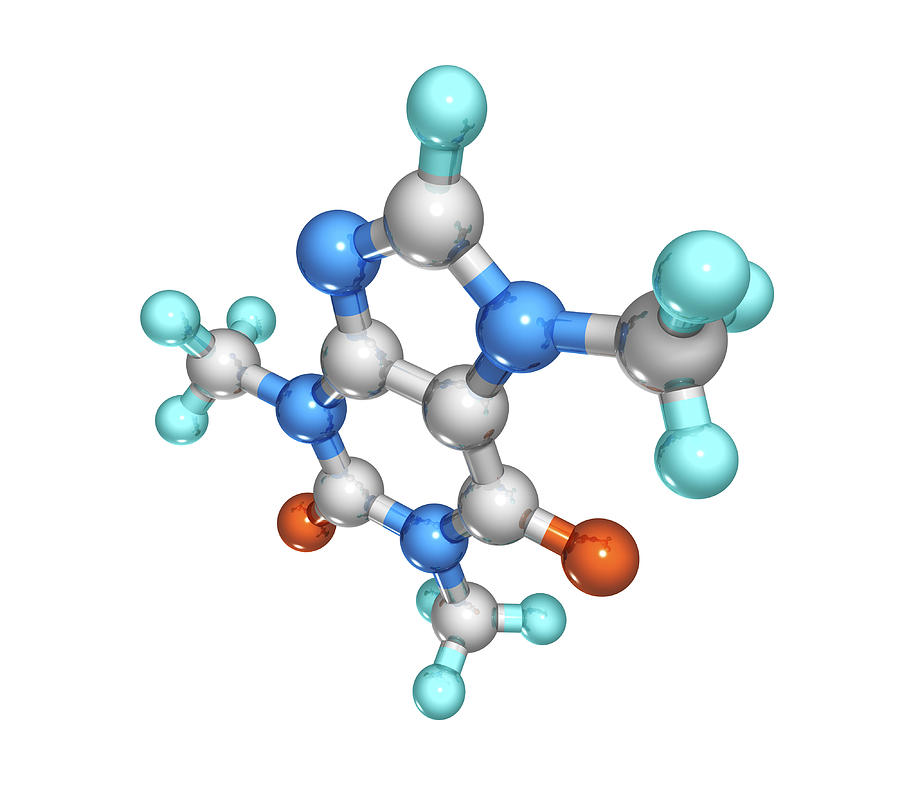
The nature of the stabilization of caffeine stacking associates. The caffeine is isolated by extraction using organic solvents and through a high-pressure extraction it is obtained a maximum possible quantity of caffeine. Simulations were performed in a cluster approximation the system contained 200 water molecules. It is calculated that tea or coffee leaves contain a maximum of 5% of caffeine. Preparation: Caffeine is mostly obtained from different plants which are cultivated with that purpose. It is also found in yerba mate leaves and guarana seeds (both of them produced in South America). The half-life (the amount of time it takes for the concentration of a substance to be halved) of. Most of caffeine is metabolized through the CYP1A1/2 enzymes into various substances such as araxanthine, theobromine, and theophylline. Occurrence: Caffeine can be found in 16 different species when the most popular are the coffee species: Coffea arabica and Coffee canephora and the tea plants. Eventually, caffeine molecules will unbind from the adenosine receptors (as all molecules generally do). Its chemical structure can be written as below, in the common representations used for organic molecules. The molecule is a typical natural alkaloid, formed by a pyrimidinedione (6-member ring with 2 nitrogen atoms) and an imidazole (5-member ring with 2 nitrogen atoms) rings which are fused. Formula and structure: The caffeine chemical formula is C 8H 10N 4O 2 and its molar mass is 194.19 g mol -1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
